ILC Cell Kit Stem Cell Research Products for Diabetes
Regenerative Medical Solutions (RMS) offers the ILC Cell Kit, a specialized stem cell research product providing pharmaceutical companies, biotech companies, and academic researchers with reliable, on-demand access to iPSC-derived islet-like clusters (ILCs) for diabetes research.
Built on over two decades of research at the University of Wisconsin and protected by a robust IP portfolio, the ILC Cell Kit is a scientifically validated cell therapy kit designed to advance diabetes drug discovery, toxicity testing, and pre-clinical research.
What Is the ILC Cell Kit?
The ILC Cell Kit is a stem cell supply containing specially formulated cryopreserved single-cell ILCs, ready for immediate use in research and drug development workflows.
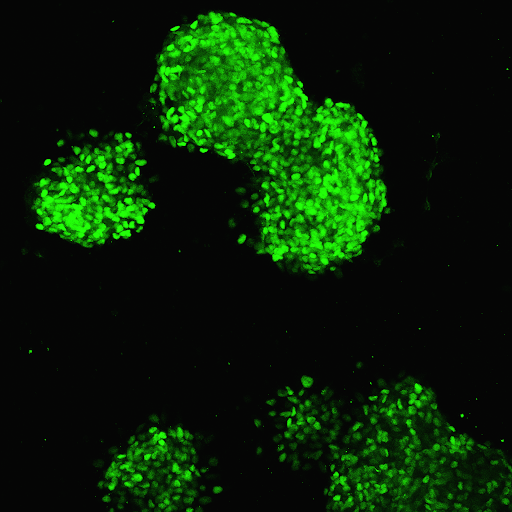
RMS ILCs are derived from human induced pluripotent stem cells (iPSCs) and closely resemble human cadaveric islets in the ways that matter most:
- Morphological similarity to human cadaveric islets
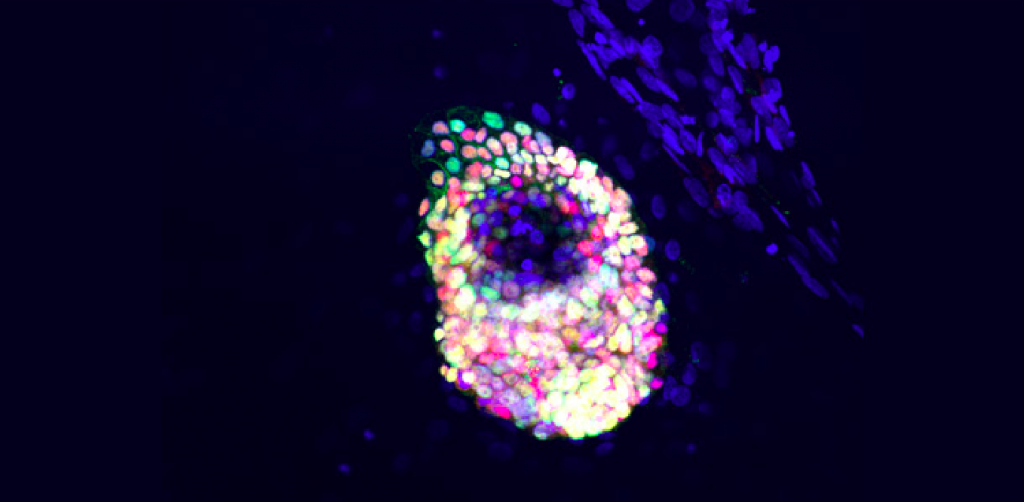
- Expression of key pancreatic markers, including NKX6.1 and PDX1
- Functional insulin production, with demonstrated glucose-stimulated insulin secretion (GSIS)
This combination of structural and functional fidelity makes the ILC Cell Kit a powerful regenerative medicine product for researchers seeking a physiologically relevant human cell model for diabetes research.
Research Applications
The ILC Cell Kit is designed for use in:
- Drug discovery: Screen candidate compounds using iPSC-derived islet-like cells that express key pancreatic markers in a physiologically relevant model.
- Toxicity testing: Evaluate the safety profile of therapeutic agents on islet-like cells in pre-clinical studies.
- Diabetes research: Study beta cell biology, dysfunction, and regeneration using a consistent, human-derived cell model.
- GSIS assays and functional studies: Assess insulin secretion responses in a reproducible and scalable system.
Advantages Over Cadaveric Islet Cells
Cadaveric islet cells have long been the standard for in vitro diabetes research, but their practical limitations are well recognized across the research community. The RMS ILC Cell Kit was developed specifically to address these challenges.| Cadaveric Islet Cells | RMS ILC Cell Kit | |
| Consistency | Donor-to-donor variability | Standardized cell quality |
| Availability | Donor-dependent | On-demand supply |
| Scalability | Limited | Scalable iPSC-based production |
| Cost | High | Competitively priced |
Our stem cell therapy supplies offer researchers and pharmaceutical developers a reliable, scalable alternative, removing the bottlenecks that slow down diabetes research and drug development.
Patented Technology from the University of Wisconsin
The ILC Cell Kit is built on patented technology developed in Dr. Jon Odorico’s laboratory at the University of Wisconsin, one of the world’s leading centers for stem cell and pancreatic research. Dr. Odorico is a world-renowned stem cell scientist and pancreatic transplant surgeon with over 20 years of experience in this field, and has previously collaborated with Dr. James Thomson, a pioneer in stem cell research.
Unlike commercially developed cell products, RMS ILCs are the product of deep, specialized scientific research. This means researchers and pharmaceutical developers are not simply purchasing stem cell supplies, they are accessing a rigorously developed, IP-protected research tool with a proven scientific foundation.
Who Uses the ILC Cell Kit?
Our regenerative medicine products are used by:
- Pharmaceutical companies developing new diabetes therapies and requiring reproducible pancreatic cell models for drug screening and pre-clinical safety studies.
- Biotech companies working on islet cell biology, regenerative medicine, or diabetes-related therapeutic development.
- Academic researchers investigating beta cell function, diabetes pathophysiology, or iPSC-derived pancreatic cell models.

ILC Cell Kit vs. ProgenMix™ Media Formulation

RMS offers two complementary stem cell research products for researchers working with islet-like cells:
ILC Cell Kit: Provides ready-to-use, cryopreserved ILCs. The ideal choice for researchers and pharmaceutical developers who need immediate access to high-quality islet-like cells without the time and resource investment of in-house cell generation.
ProgenMix™ Media Formulation: A pancreatic differentiation protocol designed for the robust generation of PDX1+ pancreatic progenitor cells and endocrine cell types. Suited to researchers who prefer to develop the cells themselves.
Request the ILC Cell Kit
To learn more about our stem cell research products, discuss your specific research requirements, or request product information, please get in touch with the RMS team.
Interested in our broader work? Learn more about our stem cell therapy for diabetes.
